In the medical device industry, compliance with relevant standards is essential to ensure patient safety, clinical effectiveness, and adherence to regulatory requirements in both local and international markets. The selection of applicable standards depends on the device’s classification, type, and intended use.
All technical standards applicable to medical devices – including those harmonized with the MDR, FDA CFR, or MDSAP requirements – are subject to periodic review and revision. The purpose of these updates is to reflect technological advancements and evolving expectations of regulatory authorities.
Standards are often updated due to:
- technological progress,
- legislative changes,
- reported issues related to the interpretation of standards,
- findings from product safety studies,
- data collected through the post-market surveillance system (Medical Devices Vigilance),
- the need to clarify specific requirements.
From the perspective of quality assurance and risk management, failure to align with the latest versions of applicable standards may negatively impact the safety of patients and users.
Additionally, it can lead to serious consequences such as:
- loss of certification,
- delays in market access,
- increased risk of complaints,
- product recalls.
Due to regulatory risks and the need to maintain compliance with current requirements, many organizations in the medical device sector choose to implement a systematic process for monitoring, evaluating, and updating the standards they apply.
Standards monitoring and change identification
A well-designed process ensures an organization responds quickly to changes, plans adaptation activities, and avoids the risk of non-compliance.
Change monitoring can be organized in various ways, depending on the scale of the organization’s operations and available resources. This responsibility may lie with a single Quality or Regulatory Affairs Specialist in smaller companies. In larger corporations, appointing a dedicated team to manage the process is common practice. The specialist responsible for the process must coordinate with the designated person responsible for regulatory compliance within the organization.
Subscriptions and memberships
The most direct method of monitoring changes to standards is subscribing to notifications and newsletters issued by standardization bodies. International and national organizations such as:
allow users to create accounts, sign up for topic-specific newsletters, or receive notifications about new publications and draft standards. Participation in technical committees and working groups additionally provides early access to information during standards’ development or revision phases. This offers an opportunity to become familiar with upcoming requirements and to influence their development.
Regulatory monitoring
Regulatory monitoring refers to the systematic tracking of changes in legislation and related documents that impact applicable standards. For companies operating in the medical device sector, this involves monitoring numerous sources – both international and local. Legislative changes may affect the content of standards and their regulatory status, such as the designation of a document as a harmonized standard.
To manage this process effectively, the organization shall assign a dedicated individual or team responsible for tracking regulatory changes. This person should regularly review communications from institutions such as:
- The European Commission – List of Harmonized Standards
- FDA – Recognized Consensus Standards
- Health Canada – Guidance Documents
- TGA Australia
Additionally, commercial systems that support regulatory monitoring can be very useful:
- Compliance Navigator (BSI)
- Emergo Regulatory Tracker – RAMS
- Regulatory Affairs Professionals Society – RAPS
These tools allow the creation of customized dashboards that filter standards by selected categories, document types, or regions. Users can configure email alerts, revision notifications, and receive expert interpretations and analyses. Such tools are especially valuable for globally operating organizations.
In addition to passive monitoring of external sources, it is worth considering a cyclical internal review of applied standards. As part of such a review:
- the list of standards used by the organization is updated,
- the availability of newer versions is verified,
- publication dates, withdrawal timelines, and transitional requirements are reviewed.
This review may be integrated with an internal audit or a Quality Management System (QMS) review.
Internal procedure for standard implementation
A Quality Management System (QMS) must include a procedure or work instruction describing the process for identifying and assessing the impact of new standard versions. This document may include:
- a definition of responsibilities for standards monitoring,
- an escalation path for communicating updates to management,
- a requirement to conduct an impact assessment,
- documentation requirements for implemented actions.
The Quality Specialist and the Regulatory Affairs Specialist are typically responsible for maintaining such a procedure. Once the procedure is in place, it is also important to provide training for new employees to ensure they understand the role of standards in company processes and know how to identify changes and report them to the appropriate personnel.
Stages of effective implementation of new standard versions
The effective implementation of a new standard version requires a planned approach, clearly defined roles and responsibilities, and a thorough analysis of the changes. This process can be divided into the following stages.
1. Impact assessment
Each new standard version should be reviewed to identify differences from the previous version. A cross-functional team should carry out this assessment, taking into account both technical and regulatory aspects.
The scope of the assessment should include:
- identification of significant changes in requirements,
- evaluation of whether the changes affect design, manufacturing, testing, documentation, or the quality management system,
- risk assessment related to not implementing the changes,
- determination of applicable transition timelines.
An impact assessment report or summary, which includes an overview of the changes, risk analysis, recommendations, and a decision on next steps must be prepared. This document must also include the assignment of responsibilities and indicate which departments must take specific actions.
2. Implementation plan
Following the impact assessment, an implementation plan for the new version of the standard should be developed. This plan should include:
- a list of actions required to implement the changes,
- designated individuals or teams responsible for each task,
- deadlines for task completion,
- the method for verifying the effectiveness of implementation,
- the method for communicating changes within the organization.
Management should approve the plan and monitor its progress. Implementation may be phased, starting with high-priority changes and followed by less critical elements. It is also advisable to allocate time for consultation with the Notified Body if the changes affect the technical documentation or certification.
3. Implementation of changes
Executing the planned activities may involve:
- updating procedures and work instructions,
- providing employee training,
- modifying product documentation,
- updating records in the quality management system,
- conducting internal audits.
Proper documentation of all actions is crucial. This documentation will serve as evidence of compliance during external audits or reviews by a certification body. Special attention should be paid to consistency across various documentation areas (e.g., procedures, work instructions, forms).
4. Verification and maintenance of compliance
Upon completion of implementation, it is necessary to verify whether all requirements of the new standard version have been met. This may include:
- conducting an internal audit,
- reviewing documentation,
- updating the declaration of conformity,
- monitoring post-implementation activities.
Maintaining compliance should be part of the continuous improvement process within the QMS. Follow-up actions such as post-implementation audits or internal surveys assessing the effectiveness of the implementation are recommended. The findings from these actions can serve as input for improving future standard implementation processes.
What should be included in the evaluation of a new standard version?
Evaluating a new version of a standard is a critical stage of the implementation process. It helps determine the scope and nature of the changes required within the quality management system. A well-conducted impact assessment enables proper planning and minimizes the risk of misinterpretation or implementation errors.
Key aspects to consider in the evaluation process include:
- Scope of changes: does the new version introduce only editorial changes, or are there significant technical modifications? New requirements, removed provisions, revised definitions, and altered approaches should be identified.
- Mandatory vs. recommended requirements: it is important to distinguish between mandatory requirements and recommendations or best practices, as this distinction affects the prioritization of actions.
- Impact on organizational processes: to what extent do the changes affect processes such as design, manufacturing, supplier oversight, or post-market surveillance? Are procedural or instructional updates needed?
- Impact on documentation: which documents must be revised or newly developed? Does the documentation update require approval from a Notified Body?
- Risk of non-compliance: what are the potential consequences of incomplete implementation? Is there a risk of non-conformity with certification, audits, MDR/IVDR compliance, etc.?
- Resources and timeline: what resources (personnel, budget, tools) are required to implement the changes? What is the realistic timeline for implementation?
- Opportunities for improvement: can the changes in the standard be leveraged to optimize processes, improve effectiveness or quality, or reduce risks?
To ensure a comprehensive understanding of the implications, the impact assessment should be conducted by a cross-functional team, including the Person Responsible for Regulatory Compliance (PRRC) and representatives from various departments. A good practice is to develop an impact assessment report documenting findings and recommendations.
Best practices for the implementation process
While each company may develop its own operational methods, several universal principles have proven effective regardless of the organization’s size or its specialization within the medical device sector.
1. Appointing a process owner
Every organization should clearly designate a person responsible for overseeing the entire process of implementing the new version of the standard. This process owner (e.g., Quality Specialist, RA/QA Manager, or Project Manager) acts as a coordinator who manages the timeline, assigns tasks, monitors risks, and resolves issues arising during the implementation.
2. Early involvement of stakeholders
Engaging all affected departments as early as possible is considered a best practice. This includes R&D, production, quality control, marketing, and clinical personnel. Early involvement allows for a more comprehensive understanding of the impact of changes and helps identify potential barriers or training needs in advance.
3. Gap assessment
Before initiating implementation, a comparative analysis should be conducted between the current quality management system and the new standard requirements. This gap assessment helps identify deficiencies and evaluate the scope of necessary changes. Documenting this analysis also provides valuable evidence during external audits.
4. Change communication
Consistent and transparent communication is one key element of a successful implementation. The project team should prepare clear communications, including summaries of key changes and their impact on daily operations. Proper communication increases change acceptance and facilitates implementation.
5. Role-specific training
Training should be mandatory and tailored to specific job roles. Operational staff should receive practical instructions related to the new requirements, while management should be equipped with tools to monitor compliance. Training should end with a knowledge test or competency verification, which serves as evidence of process effectiveness.
6. Phased implementation and iterative approach
Implementation can be more efficient if carried out in phases. For example, documentation and training updates can be addressed first, followed by changes to technical processes or IT systems. This approach allows for real-time identification of obstacles and flexible adjustment of the plan.
7. Documentation of the entire process
Each stage of the implementation must be documented. This documentation may include:
- the implementation plan,
- impact assessment report,
- action timeline,
- training protocols,
- review records,
- change logs for procedures and instructions.
Comprehensive documentation not only confirms the process’s transparency but also protects the organization from potential accusations of negligence during audits.
8. Post-implementation review
After implementation is completed, a formal post-implementation review should be conducted to evaluate the effectiveness of the actions taken. The review should assess compliance with the original plan, training effectiveness, and stakeholder engagement. Conclusions from this review should serve as a knowledge base for future implementation projects.
Template for assessing a new version of the standard
Preparing a structured assessment template for evaluating newly published standards is advisable to systematize the process of analyzing their impact. Such a document supports not only a consistent approach and comprehensive analysis but also serves as evidence during external audits. It facilitates interdepartmental communication and documents decisions and conclusions resulting from the review of changes.
This template should be used each time a new version of a standard relevant to the company’s operations is published. It can be prepared by the person responsible for the quality management system, regulatory affairs, or a dedicated interdisciplinary team. Management must approve it, and the assessment findings should serve as a basis for creating the implementation plan.
Below is an example of a template that can be used to assess the impact of a new version of a standard. It may be completed by the project team or the Quality Representative and used as a foundation for further implementation planning.
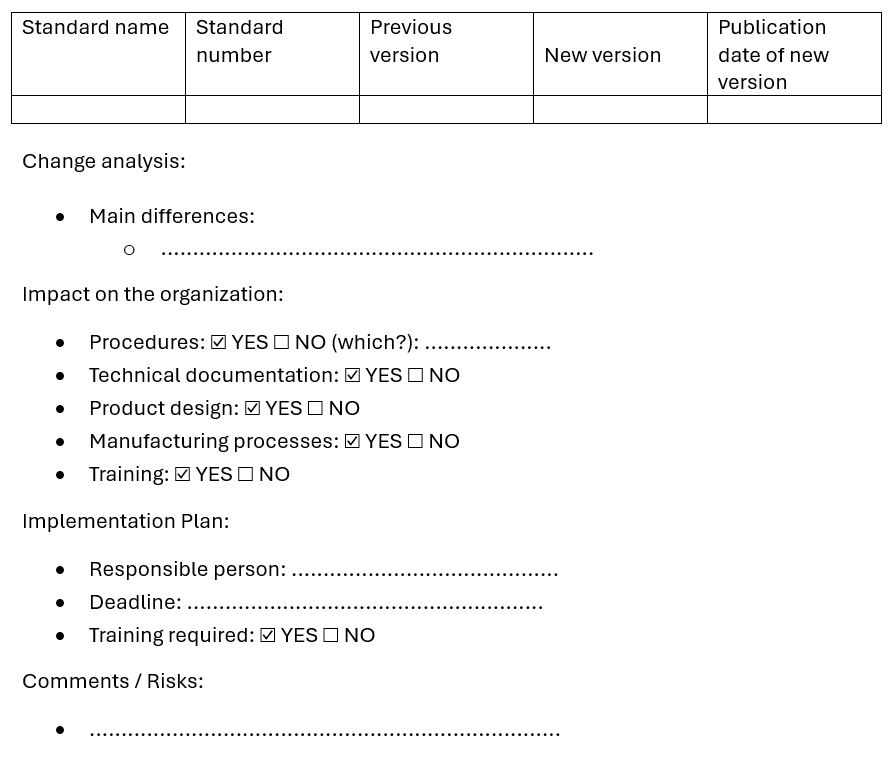

Summary
Implementing new versions of standards in a medical organization is a complex, multi-stage process that requires systematic action, an interdisciplinary approach, and strategic planning.
In particular, successful implementation requires a well-planned monitoring process that allows the organization to respond quickly to changes and prevent nonconformities. The impact assessment of a new standard version should be conducted methodically and documented to form the basis for subsequent implementation actions.
Implementation should be carried out in stages, involving appropriate teams and considering available resources. Best practices – such as conducting training sessions, updating documentation, promoting interdepartmental communication, and leveraging opportunities for optimization – significantly enhance the process’s effectiveness and added value.
It is therefore worthwhile to invest in team competence development, change monitoring systems, and tools that support the analysis and implementation of new requirements – ultimately contributing to product quality and patient safety.



















































Leave a comment